DNA Repair Group
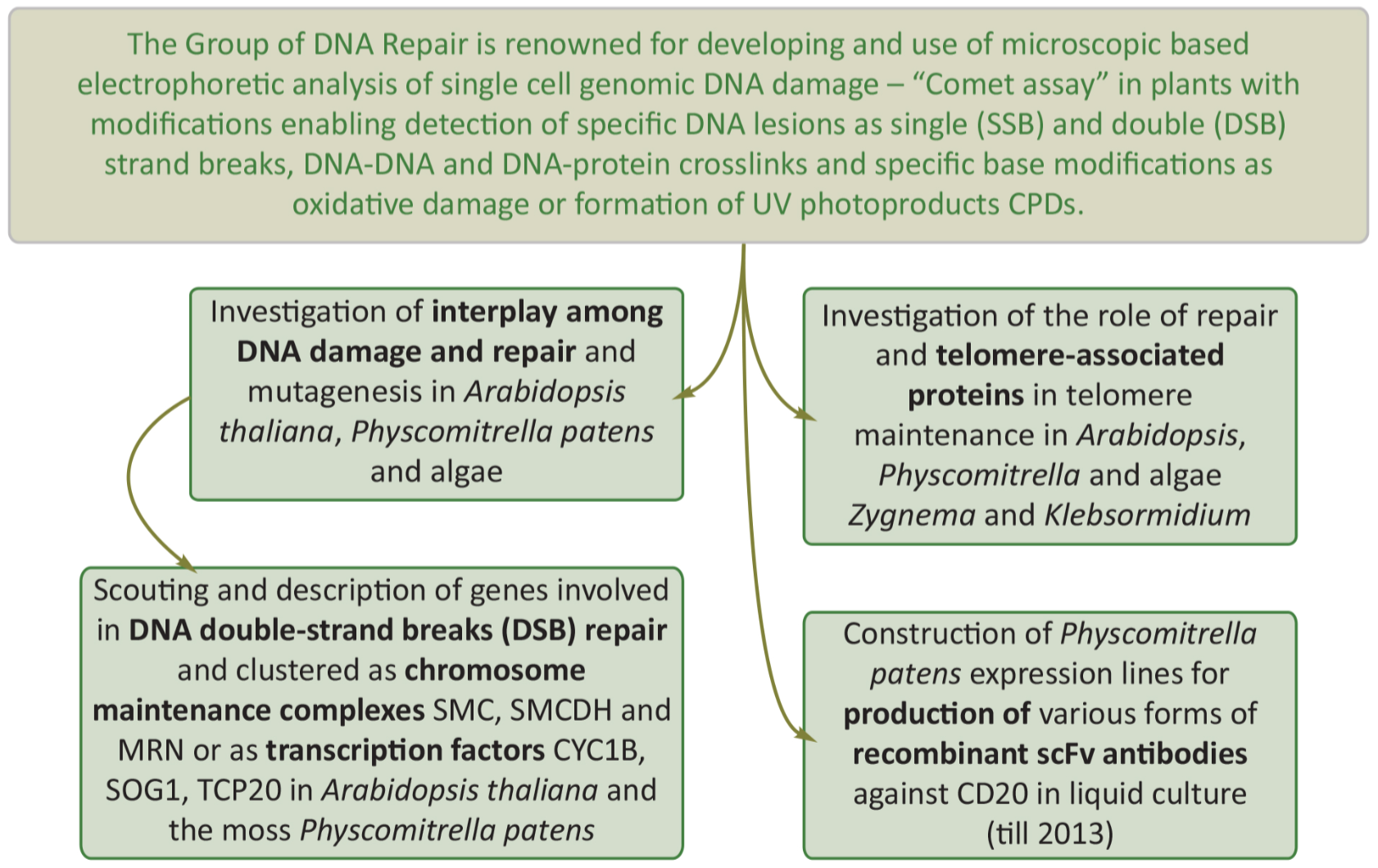
The Group of DNA Repair is world-wide renowned for developing and use of microscopic based electrophoretic analysis of single cell genomic DNA damage – "Comet assay" in plants with modifications enabling detection of specific DNA lesions as single (SSB) and double (DSB) strand breaks, DNA-DNA and DNA-protein crosslinks and specific base modifications as oxidative damage or formation of UV photoproducts CPDs. This methodological advantage of direct measurement of DNA damage (contrary to widely used indirect methods relaying on detection of transient or end products of cell response to damage like histone modifications e.g. γ-H2AX) enabled to established picture of overall two phase DSB repair kinetic with extremely rapid (so far unrecognized) first phase, which depends on Structure Maintenance of Chromosome (SMC) proteins particularly on complex SMC5/6.
SMC protein complexes form heteroduplexes that can bind sister chromatid for various purposes as SMC1/3 – chromosome cohesion or SMC2/4 chromosome condensation. Our first observation and description of novel DSB repair pathway in Arabidopsis and the important role of SMC5/6 was further investigated during 2010-2014 period.

The hurdles of using Arabidopsis model plant for DNA damage and repair study led us to acquire moss Physcomitrium patens we used for the production of recombinant antibodies. Physcomitrium besides uniquely high homologous recombination rates pose other advantages as haploid gametophyte growing in early stages in filaments. This enables by shearing to initiate culture of filament fragments 3-5 cells long, where up to 50% of apical cells are dividing.
When compared to older culture one can distinguish processes in dividing vs. Differentiated tissue. We described this approach when we in cooperation with A. Cuming, CPS, Univ. of Leeds, F. Nogue, INRA Versailles and D. Scheafer, Univ. de Neuchâtel studied moss mutants of essential DSB repair MRN (MRE11, RAD50 and NBS1) complex. Our group took advantage of haploid state and thus easy selection of mutants to study mutagenesis by positive selection of adenineribosidephosphotransferase (APT) mutants rendering resistance to 2-fluoroadenine (2FA). By combining comet and mutagenesis assays in moss enabled us to show that sensitivity of ppmre11 and pprad50 mutants to DSB induction is not due to defect of their capacity to repair them, but rather due to unrestricted high, though error-prone DSB repair leading to unsupervised mutagenesis largely representing as deletions of various sizes. This draws a picture of blocked participation of error-free homologous recombination and shifting equilibrium toward error-prone non-homologous end joining (NHEJ) pathway inducing mutations and inactivation of essential genes, thus expressing sensitive phenotype.
We explored this Physcomitrium-based combined approach to further study the role PpLIG4 and collected extensive mutagenesis data in various Physcomitrium repair background lines induced by genotoxins potentially representing various environmental stresses. Finally, in collaboration with J. Fajkus, Z. Zdráhal (CEITEC MU, Brno) and E. Sýkorová (Institute of Biophysics CAS, Brno) we are investigating the role of plant telomerase and telomerase-associated proteins in telomeres maintenance in Physcomitrium, Algea, Arabidiopsis and tobacco. The LS-MS/MS identification of candidate proteins putatively interacting with TAP-tagged TERT (telomerase reverse transcriptase – E. Sýkorová, IBP, Brno and Z. Zdráhal, CEITEC MU, Brno) has lead to the identification of candidate proteins that not only showed the likely interaction with TERT but also interesting expression profile especially in the male gametophyte. We found in Physcomitrium that telomere phenotypes are absent and DSB repair kinetics is not affected in mutants for DSB factors involved in non-homologous end joining (NHEJ). This is compliant with the overal dominance of homologous recombination over NHEJ pathways in the moss, contrary to the inverse situation in flowering plants and that algea strains Zygnema sp. 436 and Zygnema circumcatinatum TEL 181 are not responsive to DSB induction and repair at all.